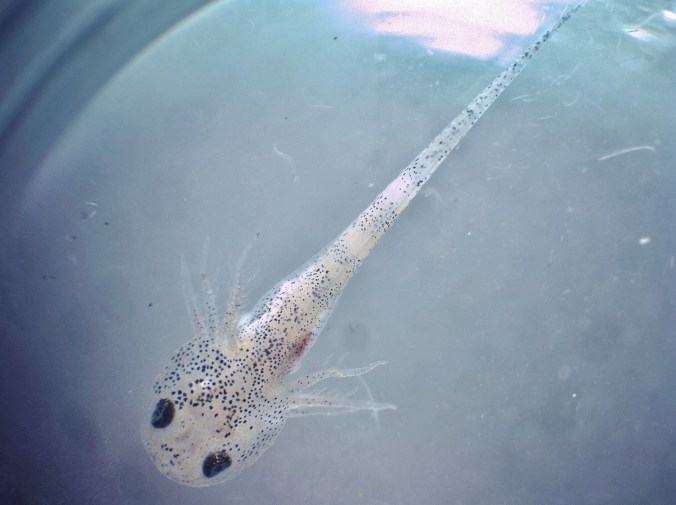
Spring is the time of year when most amphibians in the wild are emerging from hibernation, breeding, and eventually laying eggs. This pattern also holds true for the animals at the Detroit Zoo’s National Amphibian Conservation Center (NACC). Every year in the early months of spring, the top-notch staff at the NACC facilitate hibernation, breeding and rearing for endangered amphibians from all over the world with the intention of releasing them into their respective habitats in the wild. A few of these species include Wyoming toads (Bufo baxteri), striped newts (Notophalmus perstriatus) and Puerto Rican crested toads (Peltophryne lemur). Amphibians are highly sensitive creatures who rely on the environmental conditions of their native habitats to cue their natural cycles of breeding and to maintain their overall health. The NACC staff are experts at recreating these environmental cues.
Facilitating breeding in Wyoming toads, for example, is a complex process involving temperature changes and natural material for burrowing. The amphibian staff use a refrigerator to replicate the natural winter cool down experienced by these critically endangered creatures, and to ease them into a state of lowered activity and metabolism. The amphibians are also provided with a special mix of substrate to burrow into during this period – very similar to how Wyoming toads in the wild burrow below the frost line during the cold months. When they warm up after this simulated winter, the toads at the NACC are ready to breed and are paired up with mates according to the recommendations of the Association of Zoos & Aquariums’ (AZA) Species Survival Plan (SSP) for Wyoming toads. An SSP is a cooperative breeding program that is overseen by AZA-accredited institutions that breed and house animal species who are endangered or threatened.
Another example of the specialized care required to breed the unique animals at the NACC is found in the reproduction patterns of striped newts. Endemic to the long leaf pine forests of Georgia and Florida, these amphibians are known to congregate and breed in temporary pools during specific times of the year when the water levels are highest. In order to mimic these events for the striped newts at the NACC, their habitats’ water levels are lowered for a period of time – before being raised dramatically. The natural breeding cues of the striped newts are also replicated in other ways, such as changing their “photo period,” or daylight/nighttime hours, in order to simulate different seasons. Inside the animals’ habitats, the zookeepers also place specific aquatic vegetation that the newts have been known to prefer as sites for laying eggs.
Puerto Rican crested toads are among the many amphibians who are known to begin breeding activities during periods of heavy and sustained rainfall. Existing only in several small isolated populations within Puerto Rico, these toads emerge from hiding during seasonal rains to find a mate and reproduce. Making use of this knowledge, the intuitive keepers at the NACC have produced a “love song mixtape” which includes the calls of male crested toads as well as the sound of heavy rainfall. While this track may not win any Grammy Awards, it serves to stimulate the crested toads into amplexus, which is an embrace used by male toads to hold onto the female and fertilize her eggs.

After countless hours of logging temperatures, researching literature and testing water quality, the real reward comes for the staff at the NACC when these fragile and endangered animals are released into the wild having received a head start towards a brighter future. This year, in the month of June, the amphibian department at the Detroit Zoo released 3,393 Puerto Rican Crested Toads, 634 Wyoming Toads and 41 Striped Newts into native habitats ranging from the dry forests of the Caribbean Islands to the plains of Wyoming. The amphibian staff take great pride in having contributed to the conservation of some of the most important animals on the planet as well as in furthering the mission of Detroit Zoological Society.
– Mark Vassallo is an amphibian department supervisor for the Detroit Zoological Society